Abstract
Background: Bruton tyrosine kinase inhibitors (BTKis) have transformed the treatment landscapes of chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) and certain B-cell neoplasms. However, the most common mechanism of resistance is due to mutations to BTK at the cysteine binding site (C481). Nemtabrutinib (MK-1026, formerly ARQ-531) is a noncovalent, potent inhibitor of both wild-type and ibrutinib-resistant C481S-mutated BTK. Initial results from the phase 1/2 BELLWAVE-001 study (NCT03162536) showed nemtabrutinib had a manageable safety profile and promising antitumor activity in heavily pretreated patients (pts) with relapsed or refractory (R/R) CLL/SLL, including pts whose disease progressed after prior covalent BTKis (Woyach et al. Blood. 2021;138:392). We present updated efficacy for all pts with CLL/SLL treated with nemtabrutinib 65 mg and safety for all pts with hematological malignancies who were treated with nemtabrutinib at the 65-mg dose.
Methods: In this open-label, single-arm, phase 1/2 study, 9 expansion cohorts were initiated after determination of the preliminary nemtabrutinib recommended phase 2 dose (RP2D). Eligible pts with CLL/SLL were enrolled in cohort A (R/R CLL/SLL, with ≥2 prior therapies, including covalent BTKis, with documented C481 mutation), cohort B (R/R CLL/SLL with ≥2 prior therapies, intolerant to a BTKi, without C481 mutation), a dose-expansion group, or cohort I (food effect). Primary end points were ORR (per 2018 IWCLL criteria, by investigator), safety, and RP2D for pts with CLL/SLL. Secondary end points were DOR (including partial response [PR] with lymphocytosis), PFS, and safety. Efficacy analysis included CLL/SLL pts treated with nemtabrutinib 65-mg once-daily dose and safety included all pts with hematological malignancies who were treated with the nemtabrutinib 65-mg dose.
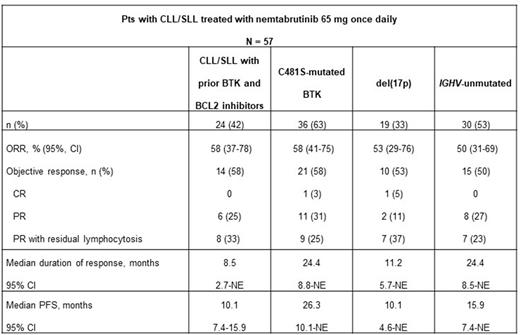
Results: A total of 112 pts were enrolled and were treated with nemtabrutinib 65 mg once daily: 57 had CLL/SLL, 46 had B-cell non-Hodgkin lymphoma (NHL), 6 had Waldenstrom's macroglobulinemia, and 3 had a diagnosis of "other.” Among the 57 pts with CLL/SLL enrolled and treated with nemtabrutinib 65 mg (cohort A, n = 25; cohort B, n = 10; dose escalation, n = 13; cohort I, n = 9); median age was 66.0 years; 16 pts (28%) were female, and 50 (88%) had ECOG PS ≤1. Median (range) number of prior therapies was 4 (1-18); 54 pts (95%) had prior BTKi therapy; 24 (42%) had prior BTKi and BCL2i therapy. In addition, 36 pts (63%) had C481S-mutated BTK; 18 (32%) had TP53 mutation; 19 (33%) had del(17p); and 30 (53%) had unmutated IGHV. Of pts with CLL/SLL, 39 (68%) discontinued, most commonly because of clinical disease progression [PD] and "other” causes (10 [18%] each); 8 (14%) discontinued owing to adverse events (AEs). Among the 24 pts with CLL/SLL who received prior BTKis and BCL-2is, 19 (33%) discontinued, most commonly because of clinical PD and other causes (6 [11%] each) and AEs (4 [7%]).
At data cutoff (April 08, 2022), median (range) follow-up for pts with CLL/SLL was 8.1 months (0.1-38.8); 32 pts had objective response (ORR, 56% [95% CI, 42-69]; complete response, 2; PR, 15; PR with residual lymphocytosis, 15). Among the 32 pts who responded, median DOR was 24.4 months (95% CI, 13.9-not evaluable [NE]); median PFS was 26.3 months (95% CI, 10.1-NE). Efficacy by key subgroups is presented in the table. Among all pts with B-cell malignancies treated with nemtabrutinib at the 65-mg dose (N = 112) included in the safety analysis, 82 (73%) had any-grade treatment-related AEs (TRAEs), most common (≥10%) were dysgeusia (21%); decreased neutrophils (20%); fatigue (13%); nausea and decreased platelets (12%, each); and diarrhea and hypertension (10%, each). Grade 3 or 4 TRAEs occurred in 45 pts (40%); most common (≥5%) were decreased neutrophils (17%) and decreased platelets and lymphocytosis (5%, each). Treatment-related discontinuations occurred in 15 pts (13%). No deaths were attributed to treatment.
Conclusion: Nemtabrutinib 65 mg continued to show promising and durable antitumor activity with a manageable safety profile in a highly relapsed/refractory population who had prior therapy with novel agents.
Disclosures
Woyach:AbbVie: Consultancy, Research Funding; Loxo@Lilly: Research Funding; Karyopharm Therapeutics: Research Funding; AstraZeneca: Consultancy; Genentech: Consultancy; Janssen: Consultancy; Newave: Consultancy; MorphoSys: Consultancy, Research Funding; Schrodinger: Research Funding; ArQule: Consultancy; Pharmacyclics: Consultancy; BeiGene: Consultancy. Flinn:City of Hope National Medical Center: Research Funding; CTI Biopharma: Research Funding; Epizyme: Research Funding; Pharmacyclics: Consultancy, Research Funding; Nurix Therapeutics: Consultancy, Research Funding; Novartis: Consultancy, Research Funding; Vincerx Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; Verastem: Consultancy, Research Funding; Acerta Pharma: Research Funding; Agios: Research Funding; ArQule: Research Funding; Celgene: Research Funding; Secura Bio: Consultancy; Xencor: Consultancy; Biopath: Research Funding; Bristol Myers Squibb: Research Funding; Roche: Consultancy, Research Funding; Unum Therapeutics: Research Funding; TG Therapeutics: Consultancy, Research Funding; CALGB: Research Funding; CALIBR: Research Funding; Takeda: Consultancy; Servier Pharmaceuticals: Consultancy; Seattle Genetics: Research Funding; Millenium Pharmaceuticals: Research Funding; Fate Therapeutics: Research Funding; Genentech: Consultancy, Research Funding; Century Therapeutics: Consultancy; Genmab: Consultancy; Gilead Sciences: Research Funding; Hutchison MediPharma: Consultancy; InnoCare Pharma: Consultancy, Research Funding; Iksuda Therapeutics: Consultancy; Janssen: Consultancy, Research Funding; Kite Pharma: Consultancy, Research Funding; MorphoSys: Consultancy, Research Funding; Incyte: Research Funding; Infinity Pharmaceuticals: Research Funding; Loxo@Lilly: Research Funding; Forty Seven: Research Funding; Merck: Research Funding; Pfizer: Research Funding; Forma Therapeutics: Research Funding; Trillium Therapeutics: Research Funding; Rhizen Pharmaceuticals: Research Funding; Portola Pharmaceuticals: Research Funding; Curis: Research Funding; Constellation Pharmaceuticals: Research Funding; Myeloid Therapeutics: Research Funding; BeiGene: Consultancy, Research Funding; IGM Biosciences: Research Funding; Tessa Therapeutics: Research Funding; TCR2 Therapeutics: Research Funding; AstraZeneca: Consultancy, Research Funding; Abbvie: Consultancy, Research Funding; Triphase Research & Development Corp: Research Funding; 2seventy bio: Research Funding. Awan:Verastem: Consultancy; Kite Pharma: Consultancy; Karyopharm: Consultancy; MEI Pharma: Consultancy; Celgene: Consultancy; Gilead Sciences: Consultancy; Pharmacyclics: Consultancy, Research Funding; Incyte: Consultancy; BeiGene: Consultancy; Johnson and Johnson: Consultancy; Dava Oncology: Consultancy; BMS: Consultancy; Merck: Consultancy; Cardinal Health: Consultancy; ADCT Therapeutics: Consultancy; Epizyme: Consultancy; Caribou Biosciences: Consultancy; Cellecter Bisosciences: Consultancy; Janssen: Consultancy; AbbVie: Consultancy; AstraZeneca: Consultancy; Genentech: Consultancy. Eradat:AbbVie: Consultancy, Honoraria, Research Funding, Speakers Bureau; Morphosys: Consultancy, Honoraria, Research Funding, Speakers Bureau; Incyte: Consultancy, Honoraria, Research Funding, Speakers Bureau; Beigene: Consultancy, Honoraria, Research Funding, Speakers Bureau; Genentech: Consultancy, Honoraria, Research Funding, Speakers Bureau; Pharmacyclics: Honoraria, Research Funding; ATARA: Research Funding; Juno: Research Funding; BMS: Research Funding; Celgene: Research Funding; AstraZeneca: Research Funding; Kite: Research Funding; Gilead: Research Funding. Brander:Ascentage (transitioning): Research Funding; Juno/Celgene/BMS: Research Funding; AstraZeneca/Acerta: Research Funding; CATO/SMS Catapult: Research Funding; NeWave: Research Funding; DTRM: Research Funding; Pfizer: Consultancy; Pharmacyclics: Consultancy, Research Funding; Genentech: Consultancy, Research Funding; BeiGene: Research Funding; MEI Pharma: Research Funding; TG Therapeutics: Consultancy, Research Funding; ArQule/Merck: Consultancy, Research Funding; AbbVie: Consultancy, Research Funding. Parikh:Adaptive Biotehcnologies: Consultancy; Phamacyclics: Consultancy, Research Funding; Janssen: Research Funding; AstraZeneca: Consultancy, Research Funding; TG Therapeutics: Research Funding; Merck: Consultancy, Research Funding; AbbVie: Consultancy, Research Funding; Ascentage Pharma: Research Funding; Genetech: Consultancy; GlaxoSmithKline: Consultancy. Phillips:AstraZeneca: Consultancy; Gilead: Consultancy; Genentech: Consultancy, Research Funding; AbbVie: Consultancy, Research Funding; Xencor: Consultancy; ADC Therapeutics: Consultancy; Beigene: Consultancy; Incyte: Consultancy, Other: Travel Expenses ; Bayer: Consultancy; Genmab: Consultancy; Eli Lilly: Consultancy; Pharmacyclics: Consultancy; Epizyme: Consultancy. Ghori:Merck & Co., Inc.: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company. Reddy:Merck & Co., Inc.: Current Employment. Farooqui:Merck & Co., Inc.: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company. Byrd:Syndax: Consultancy; Janssen: Consultancy; Novartis: Consultancy; AstraZeneca: Consultancy; Newave: Consultancy; Zencor: Research Funding; Pharmacyclics: Research Funding; Trillium: Consultancy; Ohio State University: Patents & Royalties; AbbVie: Consultancy; Kura: Consultancy; Vincerx: Consultancy, Current equity holder in private company, Current equity holder in publicly-traded company, Current holder of stock options in a privately-held company. Stephens:Newave: Research Funding; Epizyme: Consultancy; TG Therapeutics: Consultancy; Lilly: Consultancy; Genentech: Consultancy; Celgene: Consultancy; Acerta: Research Funding; Arqule: Research Funding; JUNO: Research Funding; Karyopharm: Research Funding; Mingsight: Research Funding; Novartis: Research Funding; AbbVie: Consultancy; AstraZeneca: Consultancy; CSL Behring: Consultancy; Beigene: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal